Similar Posts

Linkfest : 26 August, 2024
Some stuff I am reading today morning: Telegram app CEO Durov arrested in France ( CNBC ) Bangladesh owes $800 Million to Adani Power ( ET ) Adani’s Dharavi revamp is struggling ( Reuters ) 20 Years since TCS IPO ( BS ) New PPF & SSY rules ( Basu Nivesh ) Some thoughts on…

🏆 AURUM APEX GOLD EA 🏆🏆
Now you can download the demo version of Aurum Apex from the Comments tab to evaluate its live performance on your own broker! Aurum Apex EA is a robust 100% automated trading tool designed for the MT4 Platform. It performs real-time market analysis, detecting various trading opportunities. Suitable for traders of all levels, Aurum Apex…
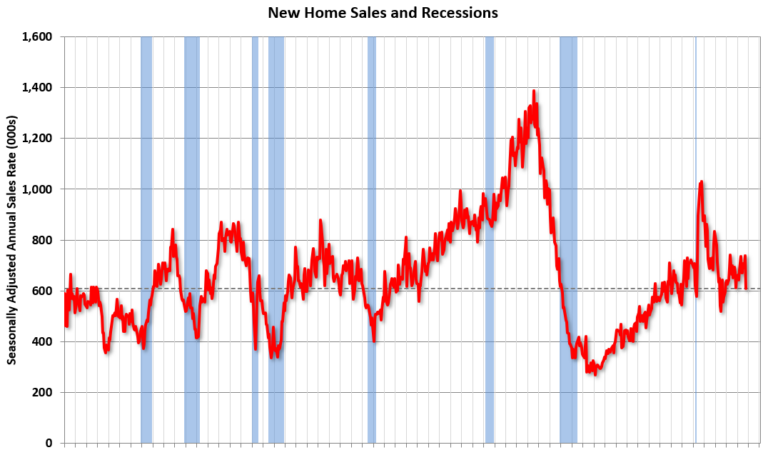
Schedule for Week of December 22, 2024
by Calculated Risk on 12/21/2024 08:11:00 AM Happy Holidays and Merry Christmas! The key economic report this week is November New Home Sales. —– Monday, December 23rd —– 8:30 AM: Chicago Fed National Activity Index for November. This is a composite index of other data. —– Tuesday, December 24th —– 8:30 AM: Durable Goods Orders…

Bonds and Yields discussion
Bond yields screaming louder than they have for a while, at Trump. Source link

Powergrid: Challenges
Source: Earnings Call of Power Grid Corporation of India Ltd Source link

Govt appoints KV Ramana Murty as Sebi whole-time member
The government has appointed Kompella Venkata Ramana Murty as a whole-time member of Sebi for a period of three years. With this addition, the regulatory board now has its full team of four full-time members. The other three whole-time members are Amarjeet Singh, Kamlesh Chandra Varshney, and Sandip Pradhan. Murty, a retired 1991 batch Indian…
