RNV – ReNerve Limited
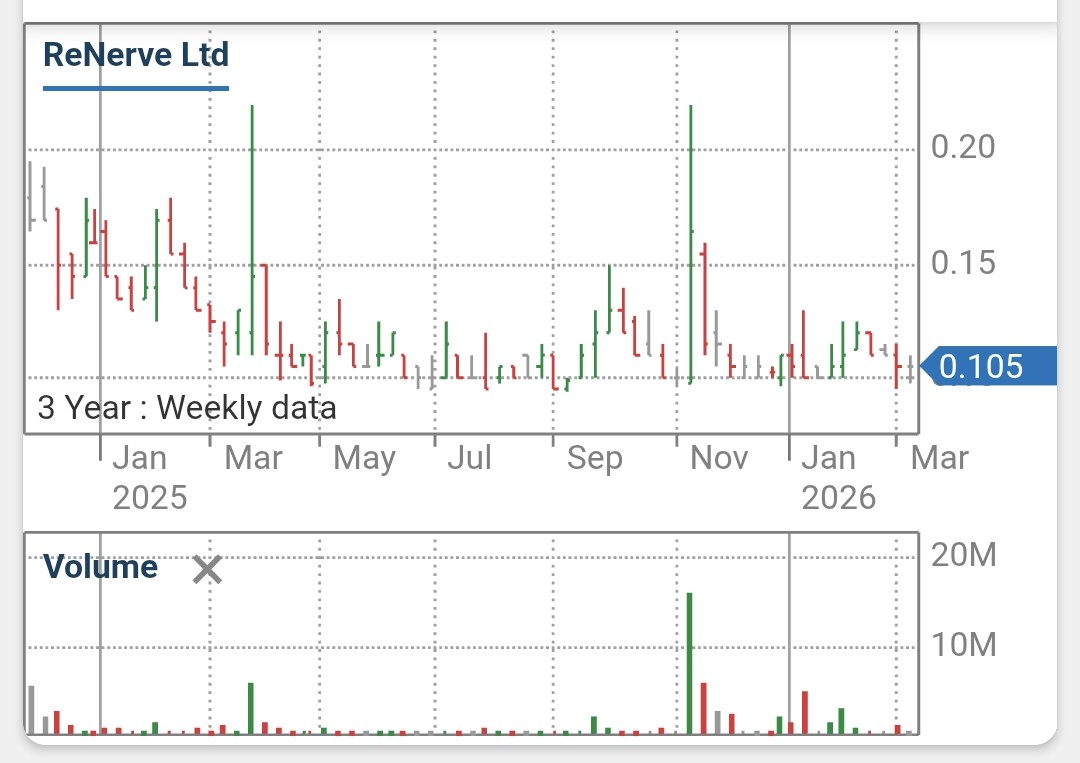
since listing
.
ReNerve releases 31 December 2025 Half Year Results
Highlights:
• Accelerating Sales Growth: In the half-year, sales increased by 60.2% in H1 FY26 compared to the corresponding period in FY25, reflecting growing adoption of the NervAlign® Nerve Cuff in the United States and increasing surgeon utilisation.
• Portfolio Expansion: Successfully launched the Empliq™ human tissue product range, achieving first sales of the Empliq™ dermal and amniotic tissue products, broadening ReNerve’s commercial offering and strengthening use of its distribution channels.
• Expanded Hospital Network Access: NervAlign® Nerve Cuff secured purchasing approvals across the US Department of Defence and Veterans Affairs healthcare systems, providing access to more than ~1,800 military hospitals, clinics, medical centres and outpatient facilities.
• International Regulatory Expansion: Secured regulatory approvals for the NervAlign® Nerve Cuff in Malaysia and Hong Kong post-period and progressed regulatory and market entry planning across additional Asia-Pacific and European jurisdictions.
• Clinical Study Progress: Continued recruitment in the NervAlign® Nerve Cuff clinical study following statistically significant interim results, with completion of recruitment expected around mid CY2026.
• Product Pipeline Advancement: Achieved key development milestones for the NervAlign® Nerve Conduit, with production hardware and manufacturing processes finalised and first production batches expected in 1H CY2026. Volume prototype production of the NervAlign® Nerve Guide Matrix was also completed, supporting planned clinical trials in CY2027.
• Closing cash of $4.18M, excludes additional $0.6m from the capital raising completed in November 2025.